 Real-Time Innovations (RTI) announced a variety of new capabilities to its industry-leading connectivity software, RTI Connext DDS, designed to advance the development of complex medical systems. Based on the Data Distribution Standard (DDS), this new product release will provide medical device manufacturers with the connectivity technology needed to address critical design challenges faced in developing secure, interoperable medical devices for healthcare systems of the future.
Real-Time Innovations (RTI) announced a variety of new capabilities to its industry-leading connectivity software, RTI Connext DDS, designed to advance the development of complex medical systems. Based on the Data Distribution Standard (DDS), this new product release will provide medical device manufacturers with the connectivity technology needed to address critical design challenges faced in developing secure, interoperable medical devices for healthcare systems of the future.
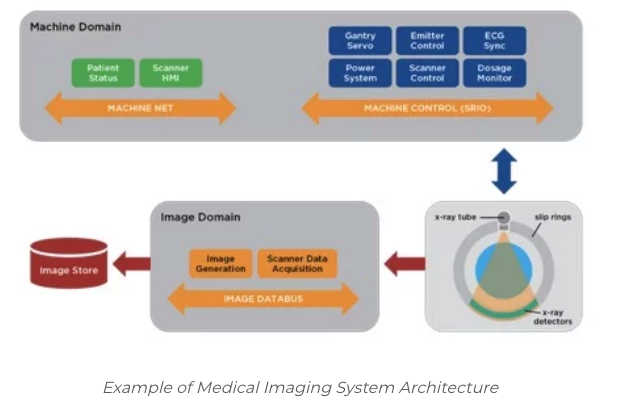
Systems of medical devices communicating seamlessly and securely have the opportunity to improve patient outcomes, reduce medical errors and lower healthcare system costs. To realize these benefits, the healthcare industry must collect and transport unprecedented amounts of data in real time, across a large number of diverse devices. For example, in the connected hospital of the future, data will not just be sent to a proprietary onsite server or cloud for analysis, but devices will share real-time data with each other securely and reliably, providing a foundation for AI and Clinical Decision Support (CDS).
Healthcare IoT systems and connected medical devices are highly complex and face a number of challenging issues throughout the design process. Developers of medical-grade products must address five major design challenges:
- Provide syntactic interoperability to lay a foundation for standards-based semantic (i.e., data model) interoperability in the future
- Ensure a secure system according to governing regulatory bodies, such as the FDA’s Guidance on Cybersecurity
- Provide access to real-time patient data from a variety of near patient medical devices
- Enable connected health solutions to scale to thousands of nodes providing critical infrastructure for device manufacturers and providers
- Provide the foundation for a highly-reliable network of devices and applications with no single points of failure
RTI Connext 6 is the first solution on the market to satisfy all of these complex, system-level requirements simultaneously, without the need to make any tradeoffs or compromises in the process. For example, developers can build their systems to scale as large as they need to without having to compromise on the level of security. This enables them to design products faster while lowering costs, and allowing them to focus on their core competencies— system development, data analysis and user experience.
In any type of healthcare environment, maintaining the highest degrees of security is of the utmost importance. The Connext DDS connectivity framework allows medical device manufacturers to secure the data in-use down to the data-type level meeting the requirements of the FDA cybersecurity guidance and minimizing the impact to system performance.
To design the connectivity software required for medical-grade systems, medical device manufacturers have the choice of designing the software in house (which is expensive, difficult and time consuming), or adopting an off-the-shelf-solution. Those that choose a well-documented, off-the-shelf connectivity framework like Connext DDS are able to meet development and security requirements, solve specific design challenges and get to market faster.
New features are compatible and interoperable across the RTI Connext product suite: Connext DDS Secure and Connext DDS Professional for standard processors and microcontrollers, Connext DDS Micro for highly resource-constrained microcontrollers and Connext DDS Cert for the most safety-critical applications
Additional new product capabilities and benefits include:
- Large Data Distribution: Allows streaming of high-bandwidth data from many disparate sources and sensors, important in both 3D imaging and Healthcare IoT applications
- Data Extensibility: Supports longevity and evolution of large, expensive medical equipment, such as large CT scanners – enabling interoperability across generations of products
- Data-Centric Gateways: Provides intelligent data routing for greater efficiency and support for integrating and scaling distributed medical systems, easing integration with legacy medical devices and enterprise
- Enhanced Performance: Reduces CPU overhead and end-to-end latency in distributed applications—a necessity for haptic control loops in surgical robotics and for the development of real time near-patent Clinical Decision Support (CDS), like in surgical robotics systems
“The lack of interoperability between devices and systems in healthcare is costing upwards of $30 billion every year and more than 200,000 lives,” said Dr. Julian Goldman, Founder and Director of the Medical Device “Plug-and-Play Interoperability & Cybersecurity Program (MD PnP). “In order to design an integrated clinical environment (ICE), the industry needs a common, standards-based platform to use as the foundation for organizations to build the ecosystems necessary to drive market-wide adoption. The Open ICE platform we have built within the MD PnP Lab is an open, proof of concept based on the DDS standard. Open ICE has been designed to provide a near patient, edge computing architecture that provides interoperability across all vendor equipment and hospital environments. Our team selected RTI’s connectivity software for its standardized data-centric approach and ability to manage secure, real-time data exchange at scale.”
New features and capabilities are available in Connext 6, generally available in Q1 2019. RTI customers can request a preview release by contacting their local sales representative.
Download a free 30-day trial of the latest, fully-functional Connext DDS software today: https://www.rti.com/downloads.
Leave a Reply